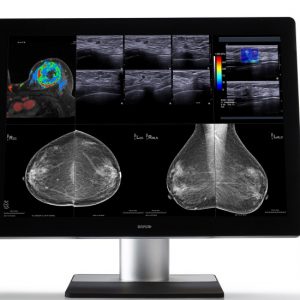
Eonis 24″ (MDRC-2224, Option WP)
- 24″ display for reviewing clinical images
- With front sensor for consistent images
- With remote QA management tools
- Fully sealed, fully cleanable
Description
This 24” Eonis® display has been built with healthcare specialists in mind. It combines consistent image quality and an attractive, versatile design with networked quality assurance. The white version display features a front glass panel and is fully sealed, making it exceptionally cleanable, even with 70% alcohol cleaning agents.
Fully cleanable design
Barco’s Eonis features toughened glass and covered cables, making the display safe to use in a clinical environment. The display has a protective front glass panel and is rated IP-32 front and back, making it easy to clean. The multiple mounting options and connections further add to the smart design.
Consistent image quality
Barco’s Eonis display has a unique front consistency sensor to automatically align the image quality every time the display is switched on. The image consistency makes collaboration between specialists easier. They can discuss images with colleagues at multiple locations, knowing that everyone is seeing identical images.
Centralized quality assurance
The Eonis display comes complete with Barco’s cloud-based MediCal QAWeb software, an online service for automated calibration, Quality Assurance and asset management. Praised in hospitals around the world, MediCal QAWeb allows healthcare IT and PACS administrators to centrally and remotely manage image quality across the healthcare organization, at the click of a button.
Features
- Front consistency sensor automatically aligns image quality
- IPS panel ensures wide viewing angle
- MediCal QAWeb suite for networked quality assurance, calibration and asset management all add to the exceptional, consistent image quality
- Exceptional cleanability with 70% alcohol based cleaning fluids (white version)
- Toughened, scratch-proof front glass
- Covered cables
- Medical-grade certifications
- Socket for Kensington lock makes it safe for use in clinical environments
- Flexible VESA mount for easy arm, wall or cart mounting
- Screen positioning in both landscape and portrait orientation
- Top connections for easy and secure connections
- Multiple inputs (HDMI, USB, etc.) ensure the display’s flexible deployment.
Additional information
| Manufacturer | Barco |
|---|---|
| Medical Display Application Type | Medical Display Monitors |
| Color | Color |
| Medical Display Resolution Options | 2MP |
Specifications
| Screen technology | TFT Color LCD IPS |
|---|---|
| Active screen size (diagonal) | 611.3 mm (24.1”) 518.4 mm x 324.0 mm (20.41” x 12.76”) (HxV) |
| Backlight | LED |
| Aspect ratio | 16:10 |
| Resolution | 2MP (1920 x 1200) |
| Pixel pitch | 0.270 mm x 0.270 mm |
| Color imaging | Yes |
| Gray imaging | Yes |
| Color support | 10 bit; 16.7 million colors |
| Viewing angle | 178° (IPS) |
| Ambient Light Compensation (ALC) | Yes, preset values in OSD |
| Front sensor | Yes |
| Maximum luminance | 300 cd/m² typical |
| DICOM calibrated luminance | 180 cd/m² |
| Contrast ratio | 1000:1 typical |
| Response time (Tr + Tf) | 14 ms typical |
| Housing color | White + Silver |
| Video input signals | VGA, DisplayPort, HDMI |
| USB ports | 1 upstream, 2 downstream |
| USB standard | 2.0 |
| Power requirements (nominal) | 100-240 V |
| Power consumption (nominal) | 40 W (less than 1.0 W in stand-by) |
| Power save mode | Yes |
| Power management | DPMS |
| Dot clock | 165 MHz |
| OSD languages | English, French, German, Spanish, Italian, Dutch, Japanese, Traditional Chinese, Simplified Chinese, Korean |
| Dimensions with stand (W x H x D) | 560.4 x 533 x 166.35 mm |
| Dimensions w/o stand (W x H x D) | 560.4 x 366 x 61.72 mm |
| Dimensions packaged (W x H x D) | 481 x 273 x 761 mm |
| Net weight with stand | 9.7 kg |
| Net weight w/o stand | 6.37 kg |
| Net weight packaged with stand | 13.7 kg |
| Height adjustment range | 100 mm +/- 5.0 mm |
| Tilt | -5° / +20° |
| Swivel | -45° / +45° |
| Pivot | 90° |
| Mounting standard | VESA (100 mm) |
| Screen protection | Protective, non-reflective glass cover |
| Certifications | CE (MDD 93/42/EEC class I product), CE-2014/30/EU, IEC 60601-1 (ED.3), IEC 60601-1 (ED.3);am1, ANSI/AAMI ES60601-1 (2005 + C1:09 + A2:10 + A1:12), CAN/CSA-C22.2 No. 60601-1 (2014), EN 60601-1-2:2015, EN 60601-1:2006/A11:2011/A1:2013/A12:2014, KC, BIS, VCCI, FCC class B, ICES-001 Level B, FDA Class I device, RoHS, BSMI |
| Supplied accessories | User Guide Cable routing strap Video cables (1 x DP, 1 x HDMI) Main cables (depending on the region of operation) USB 2.0 cable This adapter(s) is a forming part of the medical device.(Manufacturer: BridgePower Corp.; model:BPM060S12F03; Input:100-240 V AC, 50-60 Hz, 1.5 A; Output: +12V DC ( ), 5.0 A) |
| QA software | QAWeb & QAWeb Private Practice |
| Warranty | 3 years |
Digital Media
mdrc-2224-option-wp brochure